
Mark Humayun, M.D., Ph.D.
AIMBE College of Fellows Class of 2008 For extraordinary contributions to bioengineering, particularly in regards to application of an electronic device that restores vision to blind patients.
Study: Sound can stimulate blind retina to transmit signals to the brain
Via Ophthalmology Times | March 11, 2022Retinal degenerative diseases that are caused by progressive degeneration of the light-sensitive photoreceptors in the retina reman among the major causes of vision loss and blindness, affecting tens of millions of people worldwide.
Although the rods and cones which are the light-sensitive cells of the retina have completely degenerated, the neural circuitries connected to the brain are mostly well preserved providing the opportunity to restore vision by directly stimulating the retinal neurons… Continue reading.
New discovery shows long-term viability of stem cell derived retinal implant
Via Mirage News | February 4, 2022Doctors and biomedical engineers who implant cells and tissues into patients to restore motion or eyesight have long been challenged by potential rejection of the implant by the patient’s own immune system.
Now, newly published data from USC has shown a stem-cell derived implant designed to improve vision for patients suffering from age-related macular degeneration — one of the most common forms of blindness in adults over 40 — can safely be inserted into the eye and without the need for long-term immunosuppressants… Continue reading.
Researchers bioengineer first-line defense against COVID-19
Via Medical Xpress | June 30, 2020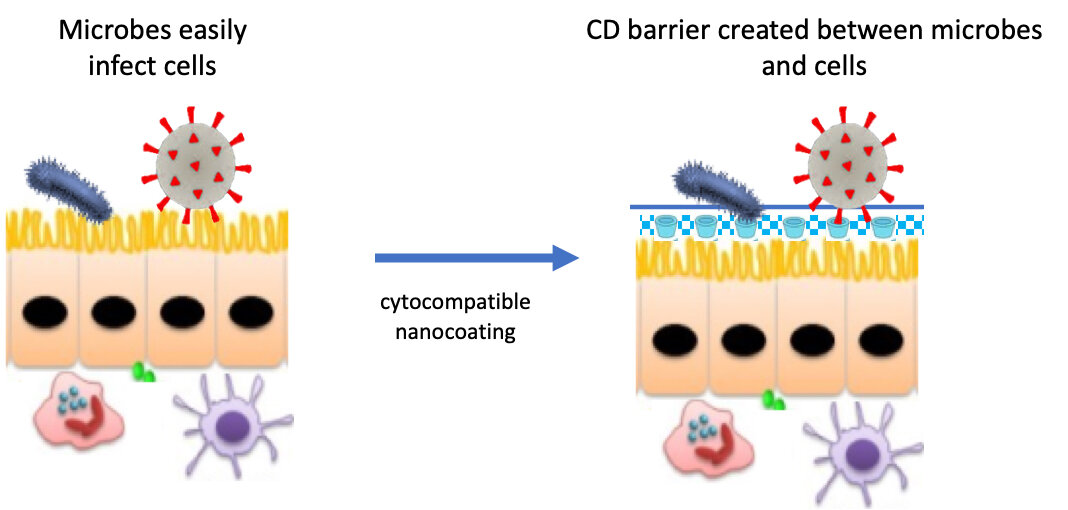
Researchers at the USC Dr. Allen and Charlotte Ginsburg Institute for Biomedical Therapeutics, the USC Institute for Technology and Medical Systems and the USC School of Pharmacy are developing an antimicrobial fluid to bolster the body’s first-line defenses against COVID-19.
The biocompatible coating is intended to block the virus from entering the body through membranes in the nose, eyes, and mouth. If successful, the invention could change the way medicine prevents certain infectious diseases.
Severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), the infectious agent that causes COVID-19, latches onto structures on the surface of our cells in order to invade. One of these membrane structures, known as a lipid raft, contains cholesterols and fats and acts as the subcellular equivalent of a dock at a seaport. Viral particles traversing our respiratory and gastrointestinal tracts hook onto lipid rafts, then enter our cells and use our cellular machinery to replicate… Continue reading.
Treatment for leading cause of blindness generates billions in value to society
Via MedicalXpress | November 15, 2019Researchers at the University of Southern California have developed an economic model to quantify the benefits of treatment for wet age-related macular degeneration (wAMD), the leading cause of blindness in western countries. Their work signals a step forward in the way ophthalmologists audit their practices to define the worth of modern treatments both to patients and society at large.
The study, led by Karen Mulligan, Ph.D. of the Sol Price School of Public Policy and the USC Schaeffer Center for Health Policy & Economics, Seth Seabury, Ph.D., director of the Keck-Schaeffer Initiative for Population Health Policy, and Mark Humayun, MD, Ph.D., director of the Dr. Allen and Charlotte Ginsburg Institute for Biomedical Therapeutics and co-director of the USC Roski Eye Institute, was published on November 14th in the Journal of the American Medical Association (JAMA)… Continue reading.
Dr. Mark Humayun elected a 2016 Fellow of National Academy of Inventors
Via PRNewswire | December 13, 2016OS ANGELES, Dec. 13, 2016 /PRNewswire-USNewswire/ — Today the National Academy of Inventors (NAI) announced Mark S. Humayun, MD, PhD, co-director of the University of Southern California (USC) Roski Eye Institute and director of the USC Institute for Biomedical Therapeutics, has been elected to the NAI 2016 Fellows Program, the highest professional distinction accorded solely to academic inventors. Humayun will attend the NAI Fellow induction ceremony at the John F. Kennedy Presidential Library and Museum in Boston next April.
Founded in 2010, NAI Fellows are nominated by their peers and chosen based on their prolific contribution in creating or facilitating outstanding inventions that have made a tangible impact on quality of life, economic development and the welfare of society.
Humayun merges medicine and engineering to focus on developing treatments for the most debilitating and challenging eye diseases and holds more than 100 issued patents and patent applications, most in the area of bioimplants for ophthalmology. Humayun is a USC professor with joint appointments in ophthalmology, cell and neurobiology at the Keck School of Medicine of USC, and in biomedical engineering at the USC Viterbi School of Engineering and holds the inaugural Cornelius J. Pings Chair in Biomedical Sciences. Humayun’s most recognized innovation is his co-creation of the Argus II (manufactured by Second Sight Medical Products Inc.), the only FDA approved retinal prosthesis system that allows those with certain blinding diseases to regain some useful vision. Earlier this year, Humayun received the prestigious National Medal of Technology and Innovation, the nation’s highest award for technology achievement, from President Barack Obama.
Mark Humayun Receives the National Medal of Technology and Innovation
Via USC | May 20, 2016President Barack Obama awarded University Professor Mark Humayun, co-director of the USC Gayle and Edward Roski Eye Institute and director of the USC Institute for Biomedical Therapeutics, the nation’s highest award for achievement in technology during a ceremony at the White House.
Humayun received the prestigious National Medal of Technology and Innovation four months after a winter storm caused the planned January event to be rescheduled.
In a press release issued by the White House, Obama stated, “Science and technology are fundamental to solving some of our nation’s biggest challenges. The knowledge produced by these Americans today will carry our country’s legacy of innovation forward and continue to help countless others around the world. Their work is a testament to American ingenuity.”
Humayun, who is one of nine recipients of the medal this year, was chosen for his lifelong dedication to bridging medical science and engineering to restore sight. He holds more than 100 issued patents and patent applications — most in the area of bioimplants for ophthalmology.
His innovative work is best exemplified by the development of the Argus II, the only Food and Drug Administration-approved retinal prosthesis system that allows those with certain blinding diseases to regain some useful vision.
“I am very honored to receive the National Medal of Technology and Innovation,” said Humayun, president elect of the American Society of Retina Specialists. “Medical breakthroughs such as the Argus II come after long periods of research and development, and I am grateful to have been and continue to be surrounded by teams of very talented individuals.”
Artificial Retina Maker Second Sight More Than Doubles In Debut
Via CNBC | November 19, 2014Shares of Second Sight Medical Products Inc, a maker of artificial retinas designed to partially restore sight to the blind, more than doubled in their market debut.
The stock touched a high of $22.45 on the Nasdaq on Wednesday, valuing the company at $777.3 million.
Second Sight’s Argus II System – the world’s only approved retinal implant – treats outer retinal degenerations, a hereditary disease affecting an estimated 1.5 million people worldwide.
The company’s net loss increased to $21.6 million in the nine months ended Sept. 30 from $17 million in the same period last year.
Second Sight offered 3.5 million shares priced at $9 each, raising $31.5 million.
The stock was up 120 percent at $19.80 after about an hour of trading, with about 1.6 million shares exchanged.
MDB Capital Group LLC was the lead underwriter for the offering.
 AIMBE
AIMBE
